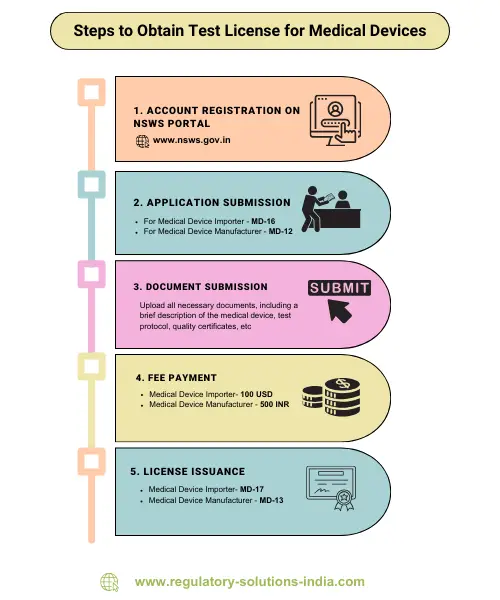
🇮🇳 India’s Plan to Exempt Low-Risk Medical Devices from Licensing: Why It Matters, Impact, and Limitations Explained
🩺 Introduction: India’s MedTech Reform for a Healthier, Faster Future
In a decisive step toward becoming a global MedTech manufacturing hub, the Government of India has proposed exempting over 1,000 low-risk medical devices and supplies from the traditional licensing process.
This reform, led by the Central Drugs Standard Control Organisation (CDSCO) under the Ministry of Health and Family Welfare, aims to simplify compliance, reduce delays, and boost domestic production of essential healthcare tools like stethoscopes, wheelchairs, surgical scissors, and diagnostic consumables.
But what exactly does this plan mean for India’s healthcare ecosystem?
Let’s break down the why, impact, limitations, and future outlook of this bold reform — and how it could reshape the Indian medical device landscape.
🔹 Why India Is Exempting Low-Risk Medical Devices from Licensing
The government’s move comes at a crucial time when India’s medical device industry, valued at over $11 billion, is poised to grow exponentially.
Here are the key reasons driving this reform:
✅ 1. Ease of Doing Business
Licensing for even basic, low-risk devices required multiple approvals under the Medical Devices Rules (2017) — a time-consuming process. Simplifying this helps manufacturers launch products faster.
✅ 2. Boosting ‘Make in India’ and Exports
India aims to reduce import dependency and strengthen local MedTech manufacturing. By cutting red tape, the country can attract more domestic and global investors to set up production facilities.
✅ 3. Global Best Practices
Several developed nations already exempt low-risk devices from extensive licensing, relying instead on self-certification models. India’s alignment with these standards enhances global credibility.
✅ 4. Improved Healthcare Accessibility
Faster approvals mean that basic medical supplies can reach hospitals, clinics, and rural health centers more efficiently — improving patient care and affordability.
✅ 5. Encouraging Innovation and Startups
Lower compliance barriers empower startups and SMEs to innovate and scale without the heavy regulatory burden that once discouraged smaller players.
🔹 The Expected Impact: A Game-Changer for India’s MedTech Industry
India’s exemption plan is designed to accelerate both production and availability of low-risk devices. Here’s how it benefits the ecosystem:
| Area | Positive Impact |
|---|---|
| Manufacturing | Simplified rules reduce cost, enabling local companies to compete with imports. |
| Regulatory Efficiency | CDSCO can focus resources on high-risk devices like implants and life-support systems. |
| Healthcare Access | More affordable medical supplies reach rural and tier-2 cities faster. |
| Startups and SMEs | Easier entry into the MedTech market encourages innovation and entrepreneurship. |
| Exports | Streamlined compliance improves export readiness and aligns India with international norms. |
This policy supports the government’s broader vision under ‘Atmanirbhar Bharat’ (Self-Reliant India) and the National Medical Devices Policy 2023, aimed at reducing import dependency (currently around 70%) and building India as a global MedTech powerhouse.
🔹 Limitations and Concerns: The Challenges Ahead
While this reform is progressive, it’s not without risks. Effective implementation will determine its success.
⚠️ 1. Classification Ambiguity
Defining what counts as a “low-risk” device requires precision. If classification is unclear, medium- or high-risk items might slip through without proper oversight.
⚠️ 2. Quality Control Risks
The self-certification model depends heavily on manufacturer honesty and robust post-market surveillance — areas where India’s regulatory system still needs strengthening.
⚠️ 3. Post-Market Vigilance
Even simple devices like syringes or wheelchairs can cause harm if poorly made. CDSCO must build real-time tracking systems for product safety and complaint redressal.
⚠️ 4. Global Export Perception
International buyers may hesitate to import “unlicensed” products unless India clearly communicates its quality assurance framework.
⚠️ 5. Need for Regulatory Capacity Building
India has a limited number of regulators per device category. As more devices enter the market, training and digital monitoring systems will be essential.
🔹 Expert Analysis: Balancing Speed and Safety
India’s reform represents a risk-based regulatory evolution — a strategy where low-risk devices get faster clearance, while high-risk devices (like pacemakers or diagnostic imaging systems) remain tightly controlled.
💡 Key Analytical Insights:
Efficiency Reallocation: By freeing resources from low-risk approvals, regulators can focus on critical, high-impact medical technologies.
Economic Multiplier: The policy can help create new jobs and encourage foreign direct investment (FDI) in the MedTech sector.
Rural Inclusion: Simplified access to devices supports healthcare initiatives like Ayushman Bharat and Health for All.
Global Competitiveness: India is positioning itself as a cost-efficient manufacturing hub similar to China’s trajectory in the 2000s.
However, to ensure credibility, India must strengthen post-market checks, update BIS standards, and digitally monitor quality certification — creating a regulatory framework that’s both agile and trustworthy.
🔹 Looking Ahead: The Future of India’s Medical Device Industry
This reform is part of a broader national strategy to make India a $50 billion MedTech market by 2030.
In the next five years, we can expect:
Integration with AI and Digital Health: Low-risk devices could connect with AI platforms for better diagnostics and patient monitoring.
Enhanced R&D and Testing Hubs: More innovation centers for device testing and validation will emerge across India.
Export Acceleration: Simplified regulation can boost exports to Asia, Africa, and Latin America.
Public-Private Partnerships: Industry collaboration will drive higher safety and quality benchmarks.
Ultimately, India is moving from being a consumer of medical technology to becoming a global creator — and this policy marks a significant milestone in that journey.
🩸 Final Thoughts
India’s decision to exempt low-risk medical devices from licensing is both strategic and visionary. It reflects confidence in the nation’s maturing manufacturing ecosystem while prioritizing healthcare access and affordability.
If implemented effectively — with strong post-market surveillance, clear classification, and global quality alignment — this reform can transform India into one of the world’s most trusted and dynamic MedTech manufacturing hubs.
As India balances speed with safety, this policy could become a model for emerging economies, proving that regulation can be both efficient and responsible in driving healthcare innovation.