1. Executive Summary: The Macro Hook
After three decades of closely watching India’s pharmaceutical journey, few companies illustrate strategic adaptation as clearly as Aurobindo Pharma. What began as a bulk drug manufacturer has steadily evolved into a global, vertically integrated formulations player with deep exposure to the US and European markets. The Q3 FY26 results, processed by the market in early February 2026, underline a critical transition phase for the company.
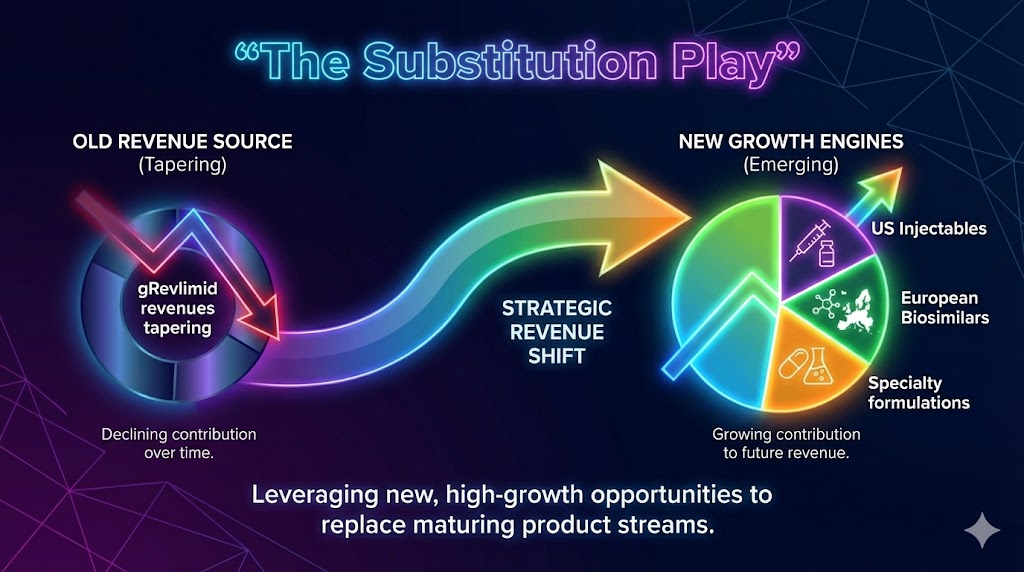
At the headline level, Aurobindo reported consolidated revenue growth of about 6% year-on-year, driven by a recovery in its US base business and sustained momentum in Europe. Net profit, however, remained under pressure, reflecting margin normalization after the gradual tapering of gRevlimid, a product that once delivered outsized profits.
The key takeaway for readers is simple: this is a bridge quarter. The extraordinary cash flows from gRevlimid are now largely behind the company. In their place, Aurobindo is building a new growth engine around US injectables, European biosimilars, and backward integration under India’s PLI framework. This shift is neither smooth nor instant, but Q3 FY26 offers early proof that the substitution strategy is working.
A crucial external factor also strengthens this narrative. The February 2026 India–US Trade Deal has reduced regulatory and tariff uncertainty for Indian pharma exporters. For a company like Aurobindo, with a large manufacturing footprint serving the US, this stability matters as much as quarterly numbers.
Aurobindo Pharma – Official Financial Results (PRIMARY SOURCE)
2. Financial Scorecard: Q3 FY26 in Perspective
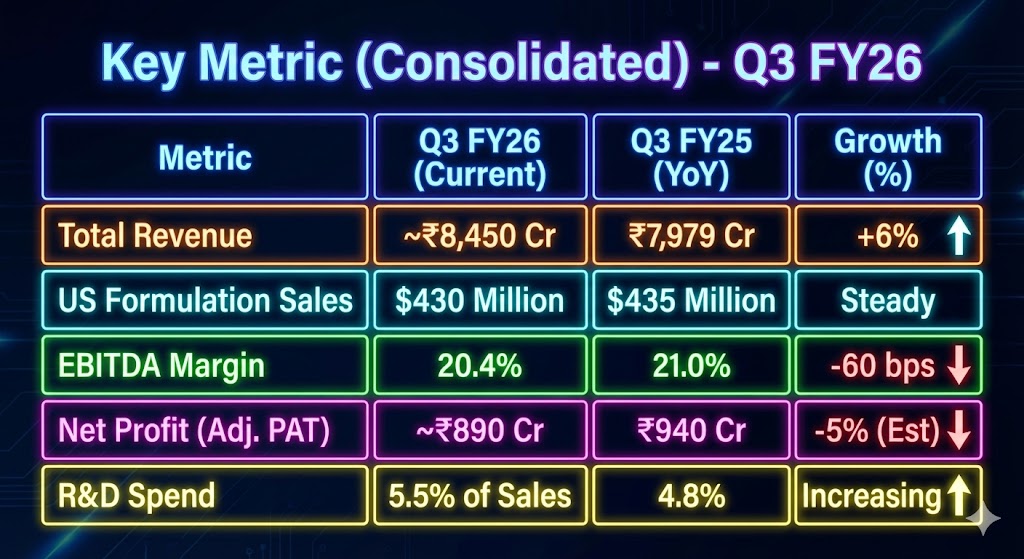
To understand the quarter properly, it is important to look at the numbers in context rather than isolation. Based on early filings and analyst estimates, Aurobindo’s consolidated revenue for Q3 FY26 stood at roughly ₹8,450 crore, up from around ₹7,979 crore in Q3 FY25, translating into 6% growth. This may appear modest compared to earlier high-growth phases, but it is meaningful given the decline in gRevlimid-linked sales.
The US formulations business, which remains the company’s largest revenue contributor, reported sales of about $430 million, broadly stable year-on-year. Stability itself is a positive signal here, as the US generics market continues to face pricing pressure and intense competition.
On profitability, EBITDA margins moderated to about 20.4%, compared to roughly 21% a year earlier. This compression reflects a mix of lower contribution from gRevlimid, higher R&D spending, and costs associated with ramping up specialty and injectable capacities. Adjusted net profit is estimated at around ₹890 crore, down slightly from last year, reinforcing that earnings are being reinvested rather than harvested.
Another important metric is R&D spend, which rose to around 5.5% of sales, up from under 5% in the previous year. For a global pharma company, this is not an expense to be minimized but a signal of long-term competitiveness, especially as Aurobindo pushes into complex generics and biosimilars.
3. Fundamental Analysis: The Three Strategic Levers
A fundamental analyst always asks where future earnings will come from. In Aurobindo’s case, three levers define the post-Revlimid strategy.
A. US Market: The Injectable Resurrection
The US remains the single most important geography for Aurobindo, but its composition is changing. While oral solids continue to face price erosion, injectables are emerging as a critical growth driver. In Q3 FY26, US injectable sales rose about 2% quarter-on-quarter to roughly $93 million.
This recovery is significant because the injectable business had faced regulatory setbacks in 2024–25, particularly at the Eugia III facility. By late 2025, remediation efforts were largely completed, and production normalized. As of early 2026, Aurobindo has regained its position as one of the largest suppliers of injectable products to US hospitals by volume.
Injectables differ fundamentally from oral generics. They have higher technical barriers, fewer competitors, and more stable pricing. This makes them ideal substitutes for gRevlimid-style super-profits, albeit with lower peak margins but much better sustainability.
The India–US Trade Deal adds a subtle but important tailwind here. With reciprocal tariffs reduced to around 18% and punitive duties removed, Aurobindo’s India-manufactured injectables are estimated to be 7–8% more profitable than in 2025, purely due to improved trade terms.
USFDA – Injectables & Drug Quality Framework
B. Europe: The $1 Billion Biosimilar Ambition
While the US market stabilizes, Europe is where Aurobindo’s next growth wave is expected. Management has reiterated its ambition to reach $1 billion in European revenues by the end of FY26, driven largely by biosimilars and specialty formulations.
Q3 FY26 marked an important milestone, with the first commercial supplies of select biosimilars entering the market. Products such as Dyrupeg, used in oncology supportive care, illustrate the shift away from commoditized generics toward complex, regulated biologics.
Biosimilars are fundamentally different from traditional generics. Development costs are higher, approval timelines are longer, and manufacturing requires deep scientific expertise. But the reward is stickier market share and slower price erosion. In Europe, where healthcare systems prioritize cost-effective biologics, this plays directly into Aurobindo’s strengths.
For readers, the implication is clear: Europe is no longer a diversification story; it is becoming a core profit driver.
USFDA – Biosimilars Program
European Medicines Agency (EMA) – Biosimilars & EU Compliance
C. Backward Integration: The Pen-G Milestone
One of the most underappreciated developments in Q3 FY26 is progress at Aurobindo’s Penicillin-G (Pen-G) plant, set up under India’s Production Linked Incentive (PLI) scheme. As of this quarter, the plant has reached around 50% capacity utilization and achieved EBITDA breakeven.
Why does this matter? Because Pen-G and its derivative 6-APA are foundational APIs for a wide range of antibiotics. For years, Indian pharma companies depended heavily on Chinese suppliers for these raw materials. By producing Pen-G domestically, Aurobindo reduces its import dependence by roughly 40% in this segment.
This backward integration is not about short-term profits. It is about supply chain security and strategic autonomy. In a world where geopolitics increasingly influence trade, such control becomes a durable competitive advantage.
4. Strategic Risks: The Watch-List
No transition is without risks, and Q3 FY26 also highlights areas investors should monitor closely.
One key issue is the integration of the Lannett acquisition in the US. While the acquisition strengthens Aurobindo’s specialty portfolio, delays in fully operationalizing laboratories or aligning compliance standards could weigh on returns in the near term.
Another factor is currency volatility. With nearly 88% of revenue coming from formulations sold largely in USD and EUR, fluctuations in the rupee can either amplify or erode margins. In early 2026, the rupee-dollar movement has been relatively stable, but this remains a variable outside management control.
Finally, while injectables and biosimilars offer better pricing stability, execution risk remains high. Any regulatory setback at a key facility can quickly disrupt supply chains and earnings.
5. Geopolitics and Policy: The MIP Shield
A major policy development in early 2026 adds a strong layer of protection to Aurobindo’s backward integration strategy. On January 29, 2026, the Indian government imposed a Minimum Import Price (MIP) on Pen-G and 6-APA.
The logic behind the MIP is straightforward: it prevents dumping of underpriced Chinese imports, which historically made domestic production unviable. For Aurobindo’s subsidiary Lyfius Pharma, this policy acts as a price floor, ensuring that domestic API production can earn reasonable margins.
This is not just a company-specific benefit. It reflects a broader shift in India’s pharmaceutical policy toward self-reliance in critical APIs, a lesson learned during pandemic-era supply disruptions.
6. Conclusion: The Value Verdict
Aurobindo Pharma’s Q3 FY26 results may not excite momentum-driven investors, but for fundamental readers, they tell a far more important story. The company has demonstrated that it can absorb the loss of gRevlimid super-profits and still grow through complex injectables, biosimilars, and backward integration.
At a forward P/E of around 14x, Aurobindo trades at a clear discount to peers such as Sun Pharma or Zydus Lifesciences. This valuation reflects skepticism about execution, but it also offers a margin of safety for long-term investors who believe in the substitution strategy.
In simple terms, Q3 FY26 proves one thing: Aurobindo no longer depends on one blockbuster product to justify its existence. It is building a diversified, resilient business model aligned with global healthcare trends and India’s evolving trade and industrial policy.
For readers seeking value grounded in fundamentals rather than hype, this quarter reinforces why Aurobindo Pharma remains one of the most interesting long-term stories in Indian pharmaceuticals.
NSE India – Aurobindo Pharma Stock & Valuation
❓ FAQ
FAQ 1
What are Aurobindo Pharma’s Q3 FY26 results?
Aurobindo Pharma reported around 6% year-on-year revenue growth in Q3 FY26, driven by recovery in US injectables and steady European business.
FAQ 2
Why is Aurobindo moving away from Revlimid?
Revlimid was a high-margin but time-limited opportunity. Aurobindo is replacing it with injectables, biosimilars, and specialty products that offer sustainable long-term growth.
FAQ 3
How important are injectables for Aurobindo Pharma?
Injectables are now a key growth driver, offering higher entry barriers, better pricing stability, and strong demand from US hospitals.
FAQ 4
What is Aurobindo’s Europe strategy?
Aurobindo aims to reach $1 billion in European revenue through biosimilars and specialty formulations with lower price erosion than generics.
FAQ 5
Is Aurobindo Pharma a long-term investment?
For long-term investors, Aurobindo offers value through injectables, biosimilars, backward integration, and policy support, though execution risks remain.